|
Last time I explained you how to collect oviductal epithelial cells, and today I would like to explain how I make my microfluidic chip which mimics oviduct to make in vitro fertilization. So first I have to design it and I have many constraints. The device has to have a membrane between two layers of PDMS (polydimethylsoloxane, a polymer). The first layer is a trap to capture oocytes and the second is just a chamber to perfuse culture medium. There is two different traps, one to capture single oocyte and another to capture multiple oocytes
|
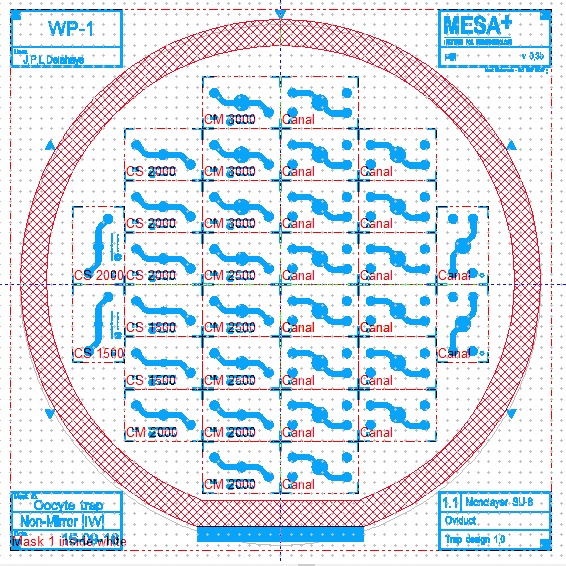
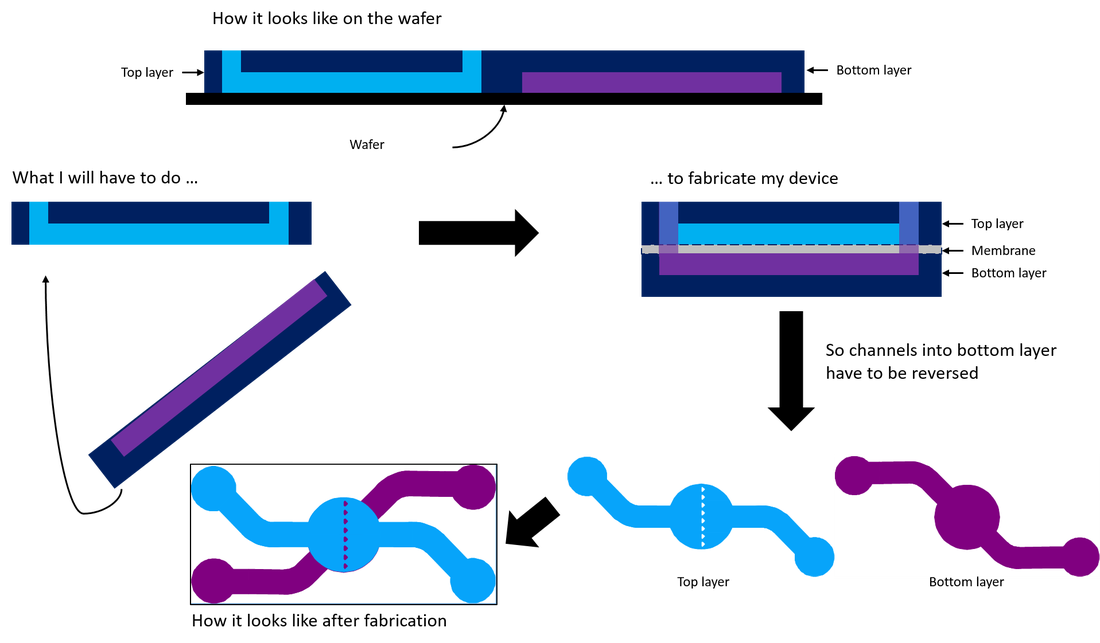
| To design my device, I used CleWin5. It is a software that looks like Paint and really easy to understand and to use. The device will use only with inverted microscope. So for this constraint, inlets and outlets have to be on the same side. I made a mark on the channel layer with the place of the inlets and outlets of the trap layer to know where punch PDMS for reservoirs. Another important thing is to thinking about the layer’s inversion for the binding because to make the device we reverse one layer to have chamber one against the other. | Design of the wafer |
Illustration of layer's inversion
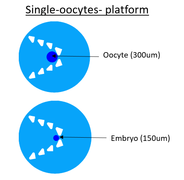
Single oocyte platform
| For the single oocyte platform, I place pillars like a V to have the oocyte on a precise position to facilitate microscopic imaging. I have two chamber sizes because we don’t know what is the more efficient in function of the secretion which is growth factors (it’s good to have a big concentration) and ammonia (it’s toxic if there is a too big concentration). So the goal is to find what is the better size to have a lot of growth factors but not too much ammonia. |
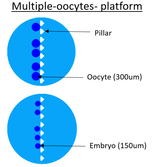
Multiple oocyte platform
| For the multiple oocyte platform, pillars are placed in line in the middle of the chamber. We suppose that each oocyte will take place between the pillars without touche another oocyte. There is three different chamber sizes for the same reason that the single oocyte trap. Sometime the quality of the embryo after fertilization is better if there is many in the same place because it increases the concentration of autocrine secretions. So the aim is to see if the single or multiple culture of oocyte/embryo change their quality. |
Pillars
| Pillars are important to capture oocyte. They have to not heart ococyte, that is why pillars have round corners. The size of bovine oocyte is 300um but the size of bovine embryo is 150um but embryo can distort itself a little bit, so the distance between two pillars have to be 100um. Than pillars are triangular because of fabrication constraints. | Pillars |
Channel layer
| The channel layer is used to perfuse culture medium. We don’t put medium in the trap layer because oocyte and embryo grow in a air interface. Nevertheless oviductal epithelial cells (which grow on the membrane in the trap layer) need nutrients to live and to secrete components for oocyte/embryo. That is why we need this layer. Here we have only one chamber size. | Channel layer |
Second steep, device's fabrication
Pourring PDMS
Polydimethylsiloxane ou PDMS is a polymere that looks like silicone. The PDMS solution is composed of 9/10 of PDMS and 1/10 of hardener. Then I put 21g of PDMS in the wafer. But before I put adhesive tape around the wafer to create border. Next, the wafer is placed under vacuum to remove air bubbles because it could disturb experiences and imaging. The PDMS is cured during one hour at 80°C. After curing, I remove the PDMS from the wafer carefully because the wafer is weak. To protect channels from dust, I put adhesive tape and I cut the pieces.
Assembly of device
The next step is to bind the two layers and the membrane. To do that, I use PDMS diluted in hexane and spin coated on glass. The aim of spin coated is to deposit uniform thin film to flat substrates. Before binding, I have to punch reservoir under microscope to be sure to punch the middle of reservoir. Then I clear layers with ethanol to remove all dust. I prepare pieces of membrane which are cut with scissor and I can begin the binding ! First, I stam top layer; secondary I put layer on the piece of membrane; third I stam the bottom layer and align the two layers under microscope and finally I cure devices 30min at 80°C under weight.
0 Comments
Hi everybody !
If you didn’t read my blog posts (it’s bad), you don’t know that my project is a partnership between the University of Twente and the University of Utrecht. If you want to know what I’m doing, read this. This is the University of Utrecht, that give me oviducts to collect oviductal epithelial cells. So during the two last weeks, I did cells culture to put them on my device, on Transwell and to freeze them. In this post, I would like to tell you the story of my cells.
Two weeks ago, I went to Utrecht to take oviducts and came back to Twente to collect cells because the laboratory of Utrecht had a contamination the week before. So it was less risked to collect cells in Twente. But I had some problem that I’m going to tell you in this post too.
If you didn’t read my blog posts (it’s bad), you don’t know that my project is a partnership between the University of Twente and the University of Utrecht. If you want to know what I’m doing, read this. This is the University of Utrecht, that give me oviducts to collect oviductal epithelial cells. So during the two last weeks, I did cells culture to put them on my device, on Transwell and to freeze them. In this post, I would like to tell you the story of my cells.
Two weeks ago, I went to Utrecht to take oviducts and came back to Twente to collect cells because the laboratory of Utrecht had a contamination the week before. So it was less risked to collect cells in Twente. But I had some problem that I’m going to tell you in this post too.
What is the solution and culture medium used ?
Before to have reproductive system, you have to prepare PBS+ and flask culture medium. The PBS+ is PBS (Phosphate Buffer Solution) with Penicillin/Streptomycin solution. It is used to wash oviduct and eliminate bacteria on the oviductal surface. The flask culture medium is composed of DMEM medium, Fetal bovin serum (FBS), Penicillin/Streptomycin solution, Gentamicin and Amphotericin B. Penicillin/Streptomycin is used against Gram positive bacteria, Gentamicin against Gram negative bacteria and Amphotericin B against fungi. We add antibiotics and antifungal because oviducts (and so the oviductal epithelial cells) come from slaughterhouse that is not clean.
I had my first problem with preparation of medium because I didn’t have Gentamicin and Amphotericin B. I had to order this but it takes some times and my supervisor told me tree day before beginning the culture that I had to make cells culture. That is why, people in Utrecht prepared for me the medium and the PBS+ that I kept in ice. So I was not good prepare whereas I could be. I really didn’t like this situation and now I anticipate what I need for the experiment and how to make it.
Where do oviducts come from ?
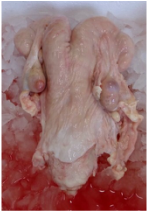
| Female reproductive system from slaughterhouse. | Oviduct comes from female reproductive system but it is not new. In Utrecht, they receive the whole female reproductive system and they have to remove oviduct from it. Reproductive systems come from a slaughterhouse in ice. It takes two hours between removing the reproductive system from the dead animal and collecting oviduct. |
How do you remove oviduct from reproductive system ?
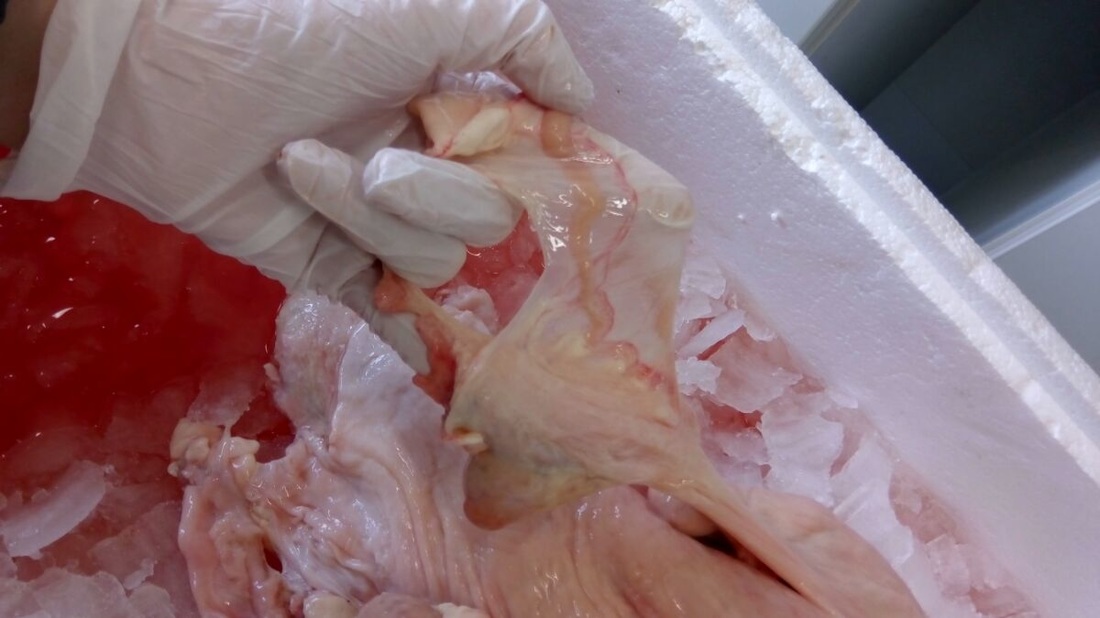
Juste find the oviduct and use a scissor! The oviduct is between the ovary and the end of uterine horn on broad ligament. Broad ligament looks like a strong translucent membrane. So to find oviduct, you have to stretch this membrane and see a little chanel which is not translucent. Then with scissor, you cut a little part of the uterine horn and and the broad ligament until the ovary. It is not necessary to be closed to the oviduct because you risk to cut the oviduct. We remove only one oviduct per reproductive system because cells in both oviduct are the same and we take more oviducts than we need in case we have a problem with one.
The next step is to remove all the broad ligament with more precise tool like a scalpel. The goal is to have a straight oviduct because it will be easier to open. Just be careful to not cut the oviduct. After removing the broad ligament, place the oviduct in 25mL of PBS+.
For me removing and cleaning oviducts are the funnier parts !
How do you collect cells from oviduct ?
Before opening oviducts, they need to be wash tree times in PBS+ to eliminate most of bacteria on the surface. Then you need to open the oviduct on a petri dish: find the fimbriae, put the scissor inside and cut the oviduct by dragging. Be careful to have always scissor inside the oviduct because it is a little bit difficult to find again the opening. You don’t have to open the whole oviduct but approximately two-thirds. Next you scrape the inside oviduct using a bladder to remove oviductal epithelial cells and place them in a falcon tube with PBS+. After collecting cells, you have to wash cells with PBS+ and centrifugation because it is possible to scrape tissues or blood cells that we don’t want. Finally place 1 million cells/mL in a flask and wait until cells are 90% confluent before use cells.
In Twente, they haven’t good scissor to open oviduct. So I didn’t open two-thirds of oviduct and I haven’t enough cells to put in flask. But it doesn’t matter, cells juste take more time to be confluent.
What are oviductal epithelial cells ?
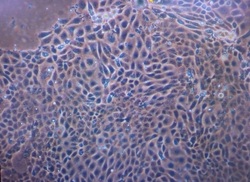
| Picture of oviductal epithelial cells in flask after 5 daysn, magnification 100x | Oviductal epithelial cells (OEC) are composed of two type of cells: cilia cells and secretory cells. In the oviduct, cilia cells beat and induce a current of oviductal fluid and move the cumulus oocyte complex into the oviduct and secretory cells synthesize and release components of the oviductal fluid. Free floating epithelial cells formed vesicles with cilia on their external surface 24h after seeding cells. First adhesion was seen 72h after seeding and after cells grow continuously. They formed a confluent monolayer after 7 days. |
Hi everyone,
I am in the University of Twente to do my internship, more precisely in the AMBER (Applied Microfluids for BioEngineering Research). My project is in collaboration with a team of the University of Utrecht (this is a city near Amsterdam) which works on in vitro fertilization.
I am in the University of Twente to do my internship, more precisely in the AMBER (Applied Microfluids for BioEngineering Research). My project is in collaboration with a team of the University of Utrecht (this is a city near Amsterdam) which works on in vitro fertilization.
But why ? Because they have a microfluidic device which is not biocompatible, so they need a device in PDMS and they don’t know how to do.
They want a structure which can reproduce the most closely real fertilization condition to improve the rate of viable embryo during in vitro fertilization and to study epigenetic questions.
They want a structure which can reproduce the most closely real fertilization condition to improve the rate of viable embryo during in vitro fertilization and to study epigenetic questions.
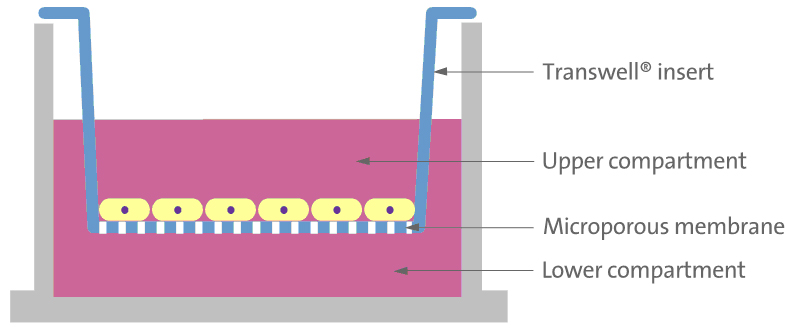
| This is the reason why, my goals are firstly to design a microfluidic device in PDMS with a perfusion system that allows holding one or multiple oocytes/embryos on a monolayer of oviductal epithelial cells where oocytes/embryos can growth, secondly to demonstrate that an oocyte/embryo can be held in position in the system, thirdly to show that a culture of oviductal epithelial cells in a Transwell insert will serve as positive control to demonstrate the potential of the cells for polarization and differentiation. | Picture1 : Transwell, system that allows to culture cells on a porous membrane (from Fitgene) |
My project is composed of different parts. I have to design the device, to create a perfusion system, to verify that the system captures oocytes and to create a monolayer of oviductal cells.
The Utrecht team give me a design brief. The microfluidic system have to be able to capture oocyte or embryo in a precise place. The system have to be closed and composed by two compartments separated by a porous membrane. I have to create two different systems, one for single oocyte or embryo and a second for multiple oocyte or embryo culture. In both systems I have to be able to implant oviductal epithelial cells. So I learn how to collect cells from the oviduct, how to culture primary cells and how to create a polarized epithelial cells monolayer.
For the design I have some fabrication’s restrictions. The thickness is 500um, so it involve that nothing have to measure less than 100um in all dimension. The system have to capture oocyte and embryo which have not the same size, so the distance between pillars have to be smaller than the embryo size because it can distort a little. Concerning the microscopy part, I have contraints too because the membrane is not transparent and the used microscope is only invert microscope.
Hi everyone !
A short post about bibliography, that could concern all people who begin a new project !
A short post about bibliography, that could concern all people who begin a new project !
When we begin a new project, each person have to stop in the square named “bibliography”. The goal is to know what had been done and used there informations for the project design. So I had during one week read a lot of articles to understand the different aspect of the topic.
| But how do you choose articles to read ? To know it, you have to dissect my subject in many little pieces ! Example: If the subject is create an oviduct-on-a-ship. So you could read about fertilization, method for in vitro fertilization, microfluidic device used for ART, organe-on-ship, co-culture of epithelial cells. |
So now, you have to search the articles and after you have to read it ! But if you have to much articles, this process could be difficult because it is not very fun… This is the reason why, you maybe need of these advice.

Do you have to read a lot of article ? What do you do to have more fun ? Have you other advice ?
This week, I had starting my internship in the AMBER team (Applied Microfluids for BioEngineering Research) in the university of Twente (Netherlands).
My project is to create on oviduct-on-a-chip that means to develop a microfluidic platform to mimic the oviduct epithelium for in vitro fertilization of bivine and/or equine oocytes followed by the embryo culture under in vivo-like conditions.
To better understand the projet, I did a lot of bibliography. Firstly I was trying to know how fertilization precisely happens and secondly to understand how to do embryo culture. In this post, I will explain what modifications happens in the oviduct during fertilization.
My project is to create on oviduct-on-a-chip that means to develop a microfluidic platform to mimic the oviduct epithelium for in vitro fertilization of bivine and/or equine oocytes followed by the embryo culture under in vivo-like conditions.
To better understand the projet, I did a lot of bibliography. Firstly I was trying to know how fertilization precisely happens and secondly to understand how to do embryo culture. In this post, I will explain what modifications happens in the oviduct during fertilization.
Anatomy and morphology of the oviduct [1]
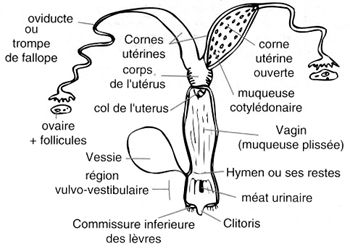
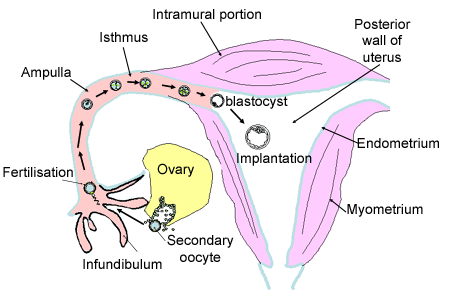
| First, I have to describe some anatomical and morphological thinks. The oviduct or Fallopian tube is a part of the female reproductive system (see picture1). It lead the egg to the uterus and it is here that fertilization happens. The oviductal epithelium is composed of ciliated and secretory cells. The role of ciliated cells is to induce a current of oviductal fluid. In the oviductal fluid, we find simple and complex carbohydrates, ions, lipids, phospholipids and proteins. It is generated by selective passage of constituents from the plasma into the oviductal lumen anb by secretion of substances synthesized by the secretory cells. Oviductal fluid reflects the conditions of the oviductal environment. | Picture 1: Schema of the female reproductive system. (from http://www.histology.leeds.ac.uk/female/oviduct.php ) |
Sperm's changes [1]
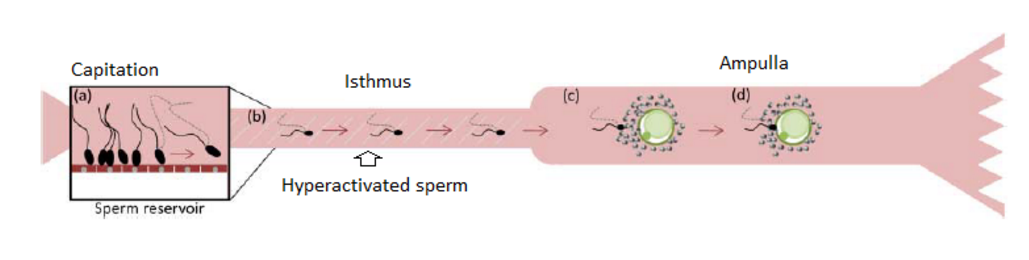
Secondly, sperm have to be modified to be able to fertilize. So after ejaculation, sperm are eliminated most in the uterotubal junction to select the best sperm which have the best motility. Then they are bound to the ciliated cells to do the capacitation. This is a physiological events that render sperm able to fertilize. Later sperm detach and acquire a new motility pattern: hyperactivated motility to be able to enter in the oocyte. Before to be hyperactivated, sperm swim straight and vigorously with symmetrical flagellar beating and after sperm swim with high amplitude and asymmetrical beating. Sperm are exposed to different microenvironment and they adapt continuously their pattern motility.
Picture2: Illustration of the sperm changes which occurs in the oviduct (from reference [1])
Oocyte's and fertilization's changes [1][2]
Thirdly, oocyte changes in the oviduct. The oviductal fluid permits the modification of the zona pellucida by molecule. Sperm can fertilize the oocyte more easily. The oocyte create thermal and chemical gradients to guide sperm in the fertilization place.
Finally after fertilization, the created embryo requires energy to develop. So the oviductal environment changes during development of the preimplantation embryo. The presence of embryo alters the expression of selected genes in the oviduct.
Finally after fertilization, the created embryo requires energy to develop. So the oviductal environment changes during development of the preimplantation embryo. The presence of embryo alters the expression of selected genes in the oviduct.
To conclude these articles, help me to understand the importance of exchanges between the oviduct and the sperm and oocyte and after the embryo. I can better imagine what can be the contraintes applied on the oviduct-on-a-chip. So now I have to know what system already exist for embryo culture to help me to design my own platform.
References:
[1] Coy, P., Garcia-Vazquez, F. and Aviles, M. (2012). Roles of the oviduct in mammalian fertilization. Reproduction, [online] (144), pp.649-660. Available at: http://www.ncbi.nlm.nih.gov/pubmed/23028122 [Accessed 11 Sep. 2016].
[2] Maillo, V., Lopera-Vasquez, R., Hamdi, M., Gutierrez-Adan, A., Lonergan, P. and Rizos, D. (2016). Maternal-embryo interaction in the bovine oviduct: Evidence from in vivo and in vitro studies. Theriogenology, 86(1), pp.443-450.