Where do chitin and chitosan come from ? Hi everyone! Today I’m going to explain you how chitin and chitosan are produced by the industries. This blogpost is based on a scientific review (ref.) describing the actual methods to extract chitin and produce chitosan.
The main source of chitin are shellfish wastes that come from several species of crustacean (crab, shrimp, and lobster). As I mentioned in previous blogpost, chitin forms a matrix with the proteins that are wrapped around. Usually, chitin is extracted following a chemical procedure. It is composed of three parts: demineralization, deproteinization and bleaching. The first is achieved by using strong acid like: HCL, acetic acid, sulfuric acid. Then, the proteins are removed employing alkaline solution (NaOH, KOH). Finally, the decolourization step is performed with bleaching agents like H2O2 or NaOCl. However, the chemical extraction have plenty drawbacks. As I mentioned in my first blogpost, it is a non-ecofriendly process because of the releasing of chemical wastes. As the chemical byproducts need specific treatments, this process is also really expensive. Moreover, this process don’t enable the recovering of proteins detached from the chitin matrix. Indeed, the use of bases and acids modify peptides structure by forming Lysoalanine and other amino acids derivatives. In this context, alternative ways of extraction have been strongly investigated. Thus, proteolytic enzymes for protein removal have been subject to many studies. Proteases are enzymes used to cut peptides bonds. So they are really appropriate to detach the protein form the matrix. The advantages of biological extraction is the recovery of peptides from hydrolysate solutions. These proteins are generally produced by Pseudomonas or Serratia bacteria Another way to extract chitin is to use chitinolytic enzymes. They are found in all biological kingdows, in both eukaryotes and prokaryotes organisms. These proteins (chitinase, chitosanase and lysozyme) cleave the bond between the N-acetylglucosamine monomers. On this way, the protein can be taking out from the chitin matrix and therefore, a crude chitin residue can be obtained. Once chitin is extracted, a deacetylation step is required to obtain chitosan. As its name suggests, in this part, the acetyl group of chitin are removed. Usually, chitosan is produced using high concentrated NaOH solution at high temperature. Nevertheless, biological extraction using filamentous fungi (Aspergillus nidulans, Mucor rouxii, Abisidia butleri) have shown very interesting results to convert chitin into chitosan. That’s it for today! I hope you found this post interesting and don’t hesitate to give me feedbacks! REFERENCE: Hayes M, Carney B, Slater J et al, Mining and marine shellfish wastes for bioactive molecules: Chitin and Chitosan – Part A: extraction methods, Biotechnol, 3: 871-877.
0 Comments
Hi everyone! I found recently an article that I used to write my internship report. I think it can be interesting for you because it is relative to my internship topic. The paper is a scientific reviews. It catalogues the numerous applications of chitin and chitosan thanks to their characteristics. I’m not going to introduce you the two polymers because I already did in previous blogpost. Nevertheless, I will explain you the state of the art concerning Industrial applications of chitin and chitosan. Because of their structure and their chemical features, chitin and chitosan are very versatile and promising biomaterials. As a matter of fact, they have a lot of interesting properties such as: biodegradability, bio-compatibility, non-toxicity and adsorption. These compounds have also high content in nitrogen (around 7 %) that make them interesting compared to other biomaterials like cellulose. Contrary to chitin, chitosan is soluble in much more solvents. That’s the reason why he has more applications. However, chitin has diverse uses. This biomaterials is employed as a raw material for man-made fibres. This fibres made of chitin and chitosan are used for wound-dressing and absorbable sutures quality. The plasters made of chitin and chitosan accelerate the healing by 75 per cent. Both chitin and chitosan are also used in water treatment. Indeed, the chemical properties of these materials is interesting to take out the heavy metal ions by chelation bounding. As chitosan is polycationic, it enables to catch anionic wastes in solution. It’s a flocculating agent. Chitin has been applied to take out plutonium and other pollutants like methyl-mercury acetate from wastewater. In cosmetics, Chitin and chitosan are widely employed in hair care, skin care and oral care. Chitosan and hair have opposite electrical charges. The first is positive whereas the other negative. Thus, chitosan is used in solution to form elastic film on hair in order to improve the strengthen and softness of hair. We found chitosan in shampoos, hair spray, hair colorants …In skin care, chitosan is used as a moisturizer (creams, pack material, lotions…). The biodegradability of chitin and chitosan is helpful in Paper industry. Indeed, they consolidate recycled paper and improve the environmental compatibility of packaging. Paper including chitosan has slicky surface and has anti-moistening properties. Microcrystalline chitin is used as emulsifying agents in Food processing. Chitin and its derivative have also been used in cell and enzymes immobilization. Chitin has digestion virtues. Indeed, the chemical is added in calves and chicken diets to improve lactose digestion. The biomaterials have applications in analytical chemistry. They are part of chromatographic support in several chromatographic separations techniques. This use, is due to the –OH and –NH2 groups that enables multiples chemical bonds. One of the key application of chitosan is in the Biomedical and Tissue Engineering field. Thanks to his tensile strength and his high permeability, many trials have been performed in order to use chitosan in artificial kidney membrane. It is also used to solve articular cartilage issues, the polymer is currently used for bone tissue, and central nervous system engineering. Finally, pharmaceutical industries employed chitosan in drug delivery systems. As it’s a huge molecule, compounds with molecular weight under 2900 kDa can pass through chitosan membrane. As you can see, these biomaterials have a LOOOOt of applications even many others that I don't noticed. The chemcial coumpounds (hydroxyl and amino groups), their biocompatibility, their size and their weight make them really interesting for industrial and research applications. There are no doubts that these materials don't reveals yet all their potentials and many discoveries about Chitin znd chitosan properties will emerge. REFERENCES:
Rinaudo M, 2006, Chitin and Chitosan: Properties and Applications, Journal of scientific and industrial research, 63:20-31. Hi everyone, I hope you are all fine! Unfortunately, I will not tell you what I’m doing at YNSECT because of the confidentiality. However, I wrote something that could interest you!
Today, I’m going to tell you how to design a good experiment. As scientists, we are all concerned by this topic. Thus, I think that it could be helpful to recall the good habits. When you are planning for an experiment, there are 4 things that you have to watch out:
Then, you have to be careful with the reproducibility of your experiment. When you are preparing samples, you have to make at least triplicates (if it’s really not possible, duplicate). It’s really important to have accurate results and to perform significant statistical analysis later. However, reproducibility doesn’t concerns only your own project. Your tests have to be reproducible by anyone. To do so, the protocol must be detailed. The material employed has to be clearly mentioned as well as the reactant used, and the settings of all the parameters. Something realy important is the control of your experiment. All the parameters implemented in your project have to be controlled (Temperature, pH, reactants concentration...).Basically, if you want to visualize the influence of a specific factor, vary this one and keep the others content. Also, don’t forget to set up negative and positive controls. Sometimes forgotten, this monitoring is really useful for response comparison. The final point to realize a good experiment is results treatment. To start, take into account all the errors incomes. When you are observing a parameters not correctly set, notice in the lab notebook. It could be an explanation for the results obtained. Then, you have to perform statistical analysis in order to give consistency to your results and to support your data. At least, standard deviation has to be calculated. If p-value, standard error of the mean or confidence intervals, is added, it will really give weight to your outcomes. To conclude, I really wanted to insist of the results interpretation. In fact, there is no bad results, and it’s not a good practices to discard the response that are not expected. What matters is to understand what you are doing. Although, it could be frustrating if results don’t match with your expectations, try to figure out what are the causes, and how to avoid it in the future. That’s it for this BlogPost! I hope that this text will be helpful and not seems too boring and repetitive, see you soon ;)! REFERENCES:
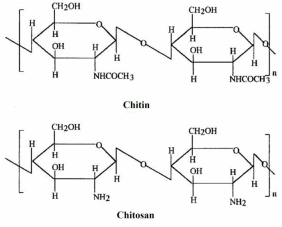
Hi everyone ! I'm here to tell about my internship at the Ynsect company! As you may understood, I'm working with insects! Before explaining the topic of my internhsip to you, let me introduce you the company. Ynsect is a new start-up specialized in agro-food biotechnologies and environment. They produce natural products from insects that are intended in several markets like: animal feed, pet food or green chemistry. The main activities are: the production of a high protein rate meal and an oil (lipids) and the extraction of chitin. The insect used is Tenebrio molitor, at the larval stage. It's usually called mealworm. Figure 1: Mealworm and adult Tenebrio: For my part, I'm focusing on the chitin extraction. But I'm not sure that everyone visualise well what chitin is! Don't worry I will explain you briefly below! Chitin is a polysaccharide composed of N acetylglucoasmine units. It's the second most abundant bio-polymer on earth after cellulose. We find it in the exoskeleton on invertebrates (crustacea, insects) but also in the cell wall of fungi. It's a very strong material. In fact, it confers a protection from predators and chocks. From chitin we can obtain a water-soluble by-product called chitosan. It's the desacetylated form of chitin. Figure 2: Chitin and Chitosan  Because of this solubility property, chitosan is used extensively in many domains like: water treatment, agriculture, cosmetics, feed, nutraceutic, biotechnology and biomedical. Usually the chitin/chitosan come from the co-products of crustacea (shrimp, lobster, crab ...). But, taking this chemical from crustacean present some disadvantages. Indeed, the shell texture is not the same all over the year, it oscillates with season. Moreover, it contains minerals that have to be removed with strong acid and bases. Finally, the shellfishes are expensive and need water treatment to be breed in good conditions. Insects don't have all of those constrains. Their shell don't have a lot of minerals, their chitin has the same consistance all over the year. Besides, they are cheaper and easy to bred. Figure 3: The chitin matrix As you can see on the picture, chitin forms a complex with protein which are wrapped around the polysaccharides. To obtain crude chitin, we have to remove those proteins, that's why we use specific enzyme called protease. This kind of enzyme is specialized in the hydrolysis of peptidic bonds. Usually, the chitin is extracted by a chemical way (several reactions using a lot of sodium hydroxyde). With this method, the yield is great but it has several drawbacks: - the chemical reactions releases a lot of by-products those are difficult to recycle. - the sodium hydroxyde at high concentration can damage the chitin - It doesn't allow to recover the protein wrapped around chitin So my work consists on assaying to obtain a high quality chitin, the purest, in a more sustainable way. That's it for my internship overview. I hope that you enjoy by reading it! See you soon for another blogpost! REFERENCES:
|