|
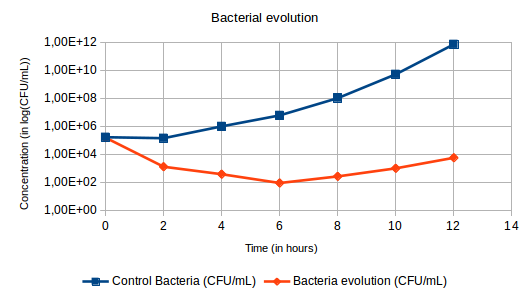
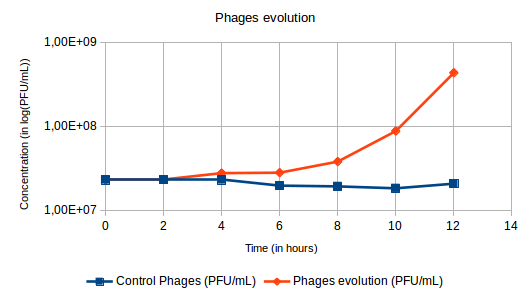
Hi everyone ! :) In a precedent post, I spoke about phage therapy but I didn’t explain you what it was at that moment. So I will explain you now. The process of phage therapy is simple. Its consists in the use of bacteriophages, more commonly called phages to infect and kill targeted prokaryotes (bacteria and archaea). Phage therapy has many advantages in comparison with the conventional methods. In fact, phage therapy can target specific bacteria ; phage-resistant mutants are less frequent than bacteria resistant to antibiotics ; it has a lower environmental impact; phages are easier licensed for fisheries use than antibiotics ; it is a fast and cheaper method ; and phages are more resistant to environmental changes conditions. However, phage therapy requires knowledges of the concentrations and times of application of bacteria and phages, and of all the interactions factors with the environment. Besides, phages multiply themselves by infecting bacteria (See the picture of the lytic and lysogenic cycles from my precedent post “A bacteriophages project : Welcome to the ENVIPHAGE Project” from the 15/10/2016). To do experiment on phage therapy, first we have to know the concentration of bacteria after a certain amount of time after reviving the bacteria. So the first day we have to revive the bacteria overnight and the day after we have to take 100µL of bacteria and but in 30mL of TSB (liquid medium rich in nutrients for the development of bacteria) for 10 hours. After these 10 hours you check the concentration of bacteria using the Poor Platting technique. It consist in putting 100µL of bacteria on the middle of an empty plate and adding 10mL of TSA. For the phages’ title we have to use Double Agar Layer technique. We dilute the stock that we produced or that we already have and put 200µL of bacteria in TSA soft (a semi solid medium) with 500µL of dilute solution and we spread all of it on a plate with TSA (solid medium for the development of bacteria in plates) and put in the incubator. After having the concentration of bacteria and the phages’ title we calculate the volume after 10 hours of development to put in 30mL of TSB to obtain a concentration of 105 bacteria/ mL. For the volume of phage, we calculate the volume to put to obtain a ratio of 10, 100 or 1000 in function of the bacteria. Since we know the volumes to use for our experiment, we put these volumes in 3 erlenmeyer with 30mL of TSB : one with only bacteria which will be the Control Bacteria ; one with only phages which will be the Control Phages ; and one with bacteria and phages where the process of phage therapy will occur, called Bacteria + Phages. The experiment takes place during 12 hours and every 2 hours bacteria and phages must be quantified. Generally, for the first time we do this experiment we have to use a large range of dilution because we didn’t know yet how the phage we have would work. So we dilute following that program : For the Control Bacteria : - T0 : 10-1, 10-2 and 10-3 - T4 : 10-4, 10-5 and 10-6 - T1 : 10-1, 10-2 and 10-3 - T5 : 10-5, 10-6 and 10-7 - T2 : 10-2, 10-3 and 10-4 - T6 : 10-6, 10-7 and 10-8 - T3 : 10-3, 10-4 and 10-5 For the Control Phages : - T0 : 10-5 and 10-6 - T4 : 10-5 and 10-6 - T1 : 10-5 and 10-6 - T5 : 10-5 and 10-6 - T2 : 10-5 and 10-6 - T6 : 10-5 and 10-6 - T3 : 10-5 and 10-6 For the analysis of the phages in the Bacteria + Phages : - T0 : 10-5 and 10-6 - T1 : 10-5 and 10-6 - T2 : 10-5 and 10-6 - T3 : 10-5 and 10-6 - T4 : 10-5 and 10-6 - T5 : 10-5 and 10-6 - T6 : 10-5 and 10-6 For the analysis of the bacteria in the Bacteria + Phages : - T0: 10-1, 10-2 and 10-3 - T1: 10-1, 10-2, 10-3 and 10-4 - T2: 10-0, 10-1, 10-2 and 10-3 - T3: 10-0, 10-1, 10-2 and 10-3 - T4: 10-0, 10-1, 10-2 and 10-3 - T5: 10-0, 10-1, 10-2 and 10-3 - T6: 10-0, 10-1, 10-2 and 10-3 For the Control Bacteria and the analysis of the bacteria in the Bacteria + Phages, we use the Pour Platting technique, and for the Control Phages and the analysis of the phages in the Bacteria + Phages we use the Double Agar Layer technique. Expected results : We expect for the concentration of the bacteria to increase for the Control Bacteria because the bacteria are in a nutritive medium without phages and to decrease for the Bacteria + Phages because the phages are killing the bacteria (Figure 1). For the Control Phages, we expect for the title of phages to remain constant because the phages cannot multiply themselves without bacteria and for the title of phages of the Bacteria + Phages to increase because they multiply while killing bacteria (Figure 2). Figure 1: Expected results of the evolution of the bacteria's concentration during phage therapy. Figure 2: Expected results of the evolution of the phages' title during phage therapy. If you have some question about phage therapy, it will be a pleasure for me to answer you.
See you soon ! Nicolas
0 Comments
Hi everyone !
I am in the writing of my internship report and an interrogation came to me about the reference. When you are doing some background researches for your introduction or even for your own comprehension, can you insert any you want sources ? I explain : You insert an information and you add a source to that information. But the source is old, about 25 years old. Must you find an other source, more recent, to corroborate that information ? Assuming of course that in your field 25 years old is long. I means in some subjects it might be more or less short period of time. Until when an article can be quote in a term of time if other more recent articles can give the same information ? And I was talking with some PhD students and they told me that they can’t quote an other thesis in their own thesis. Is it the same where you are ? See you ! Nicolas Hello everyone !
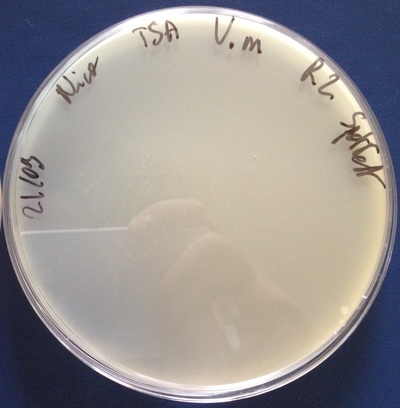
I hope you are all fine. In my last post I told you that I will explain you how to text the efficiency of a specific strain of bacteriophage to inactivate bacteria. Unfortunately, The experiment went wrong so I will wait to have a successful one to try to explain why it went wrong and to present you the results of multiples technical and biological replicates. So finally, I will speak here about how I feel about my internship and the tips and tricks that I gain since the the start of my work two months ago. First of all I want to join Loïc on the fact that going to thesis defenses bring a lot of information [1]. But if you are unfortunate like me and that the PhD students of your laboratory already have done their defense or that they will do it when you won’t be their any more, you feel unlucky ! But don’t worry ! You still have a way to access to theses knowledges ! In fact, if you have PhD students that have already done their defense you can ask for their thesis statement ! I can agree and understand that for a night reeding before sleeping a thesis can be a bit hard but it is full of plenty type of information, diagrams and protocols that can interest you ! Indeed, in a thesis defense, the PhD student can speak about everything because he or she have a limited time. So reading their statement to find more details information can be very useful ! Besides, even if the work inside could be to much difficult to understand for you, you have still the opportunity to ask further question about it at the laboratory the next day ! And I am speaking a lot about PhD students but Master students can bring a lot of help too ! Before doing this internship, I had the general idea that “Yeah, this internship will help me to see how is the real work in a laboratory” ! But now I am more in the mood that “Yeah this internship combine with the experience that I already have will help me to improve my work in a laboratory” ! Since we already have worked in laboratories we know a bit how to work in a laboratory. But the most important thing in an internship, without speaking about the knowledges you will gain, are the tips and tricks that you will take back from it. In every laboratories you have some “ways of doing a thing” that could be better that the actual way you learn or worst. So you just have to improve your work AND the work of the lab ! In fact, you have to learn the better way and teach some of the tips and tricks that you know and which can improve the work of your colleagues. Because an internship can’t be only beneficial for you ! For example, here in the laboratory where I work, we have burner inside the hood, which can help you to sterilize some bottles or even some eppendorfs and can give you a more sterile environment to do experiment. Besides, using alcohol to sterilize your hands and the hood are good, but what for the eppendorfs boxes or tips boxes or even micro pipettes ? Or even the support to put eppendorfs and falcons ? Morality : If you want to avoid contamination in your work, don’t forget to put alcohol on every thing that enter in the hood. See you soon for my next post ! :) Nicolas References : [1] http://stagelfdv.weebly.com/home/-conferences-and-thesis-defenses-a-gold-mine-of-informations-loic Hi everyone ! I hope you are all fine. Today I will introduce you what I did for the previous two months and what will be my work for the next one. As I said in my precedent post, one f the biggest part of the project is to experiment on the bacterial inactivation by bacteriophages. The process is very simple : You have to see how efficient a phage target a bacteria. But before that, it means that you have to find the phages ! Since we are in a coastal city, we are able to search phages directly in the water and not just by them to an other laboratory that already have phages for the targeted bacteria. There are a lot of pathogenic bacteria that involve problems in fish and shellfish farming, but with the increase of the temperatures of the oceans due to the global warming make the bacterial family of Vibrio [1] and Aeromonas [2] very dangerous [3][4]. In fact, according to the Centers for Disease Control and Prevention, Vibrio is known to cause “80,000 illnesses and 100 deaths in the United states every year” [5]. So It was decides that I will work with three different type of Vibrio which are Vibrio parahaemolyticus, Vibrio anguillarum and Vibrio metschnikovii ; and one type of Aeromonas which is Aeromonas hydrophila. The phages isolation is a long process which can take days. In fact, as a lot of things in biology it requires time for the bacteria and phages to multiply. The process is very simple. The first thing you have to do is to take water samples from the see or the river next to the see when the tide is high, or sewer water sample too. After that you take bacteria that you already put to multiply the day before and you mix the water sample (filtrated or not, it depends of what you want), the bacteria and a media very rich in nutrients for the bacteria. After letting incubate overnight, you have to do a spottest. The spottest is a very simple way to see if you have pages in your sample. You take a plate whare you spread a high concentration of your targeted bacteria and you put on the middle some drops of your liquid that you had already centrifuge to separate the phages from the bacteria. If the spottest is negative, you have to start again all the experiment. But if the spottest is positive, congratulations : you have phages ! (See figure bellow) Example of positive spottest Example of negative spottest All you have to do after that is to find the concentration of you stock. I was supposed to do it all my internship, but fortunately I handle this part quietly and after two months and a lot of failure in my search of phages, I finally found and produce a stock of bacteriophages for the Aeromonas hydrophila ! So I will continue to search phages, but I will do in the same time experiments to evaluate the efficiency of the phages I found. But I will do this experiment only next week so I ill explain it and my next post. See you soon ! :) References :
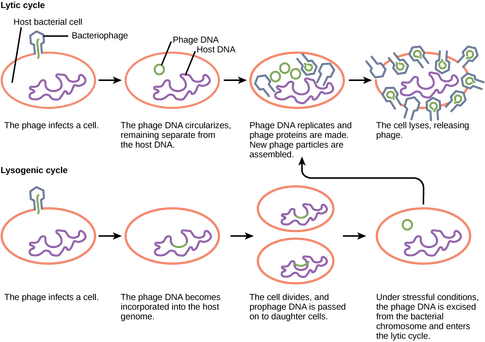
[1] https://en.wikipedia.org/wiki/Vibrio [2] https://en.wikipedia.org/wiki/Aeromonas [3] As ocean temperatures rise, so too will Vibrio outbreaks, Clare Leschin-Hoar [4] Studies on Aeromonas hydrophila in Cultured Oreochromis niloticus at Kafr El Sheikh Governorate, Egypt with Reference to Histopathological Alterations in Some Vital Organs, A.E. Noor El Deen, Sohad, M. Dorgham, Azza H.M. Hassan and A.S. Hakim [5] https://www.cdc.gov/vibrio/ Hi everyone ! Since already a month, I began my internship one of the biology laboratory of the University of Aveiro in Portugal, where I joined the ENVIPHAGE project [1]. The ENVIPHAGE project involves Spanish research and development bodies (Fundación AZTI and Biopolis SL), a research group from the University of Aveiro (Portugal) and Aguacircia fish farm (Portugal). The main purpose of this project is to evaluate the impact of bacteriophages use in environmental bacterial ecology. Picture from GoogleMaps Why this project ? Over the years, we noticed an increase in the world total aquaculture production. In fact, this production has increased from 8.7 million tons of fish in 1990 to 31.6 million tons in 2006 [2]. But that increase wasn’t without any consequences ! The number of pathogenic infection in the fish farming increased too. To face the problem, scientists and companies tried to take preventive measures to limit the problem, for example, the use of antibiotics. But there are several factors that accentuated this serious problem, such as the very variable environmental conditions [2] ; the resistance against antibiotics in common pathogenic bacteria [2] ; the destruction of the bacterial population the the environment [2] ; and the few licensed antibiotic for fisheries uses [2]. But since a few years, an alternative to antibiotics have been tested : the bacteriophages, also called phages. What is a bacteriophage ? Phages are viruses which infect targeted species or strains of bacteria. It exists 2 types of phages, lytic and lysogenic [3]. (See the picture bellow to understand the two different types of phages) Picture from http://cnx.org/resources/a0861053f05c94a3b8275aef46686538e3e073d2/Figure_21_02_03.png Since phages are supposed to inactivate only targeted bacteria, this alternative could avoid problem such as the destruction of the bacterial environment. Besides phages are viruses, so they have a higher rate of mutation than the bigger part of the bacteria and that make them more efficient to face some bacterial mutation and avoid resistant bacteria. But concretely, what is the goal of the project ? The first objective of this project was to test if phages could be use in real condition to fight against pathogenic bacteria. The team finished that part before my arrival. I will talk more about that part in an other post. The second objective is to see if the pages that we could possibly use in fish farming would make any environmental damage or not. See you soon for a next post, where I will explain what I am doing there. References :
[1] http://www.enviphage.eu/en/ [2] Phage Therapy and Photodynamic Therapy: Low Environmental Impact Approaches to Inactivate Microorganisms in Fish Farming Plants, Adelaide Almeida, Ângela Cunha, Newton C.M. Gomes, Eliana Alves, Liliana Costa and Maria A.F. Faustino [3] Bacteriophage remediation of bacterial pathogens in aquaculture: a review of the technology, Gary P Richards |