|
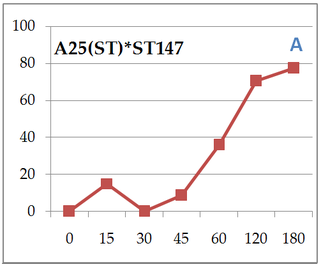
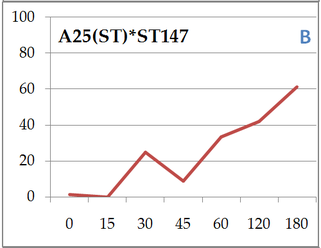
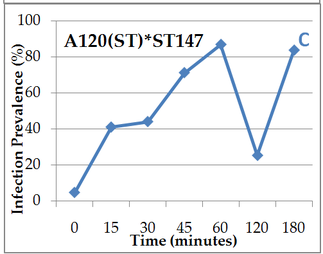
Everybody hello! I must say that I’ve been quite busy those 2 last months organizing night follow experiments on dinospores’ fate. Working in a laboratory implies protocol reviews and team management, which has been a big and great practice during past weeks! So here we are, after calibration and good maintaining of targeted cultures I finally started. Table 1. gather all cross-infection I worked on (for further understanding, refer to pasts article: An idea for Amoebophrya’s dinospores maturation time?). Experiment 1: Optimisation of incubatin time First part of the protocol was to obtain details on how long both parasite strains need to be in contact to initialize an infection in ST147 (Scrippsiella trochoidea). I designed a protocol based on previous results (refer to: An idea for Amoebophrya’s dinospores maturation time?) that have shown that maturation time for dinospores of strain A25(ST) is around 18h and 6h for A120(ST) in host ST147. '>  Table 1. Incubation time for strains A25(ST) and A120(ST) to initialize host infection The minimal time required for infection was checked using incubations from 15 minutes to 3 hours. Step 1 consisted on disnospore production: filter cultures onto 5 µm polycarbonate filters and inoculated on a new fresh host ratio 1:10. Step 2 consisted of collecting dinospores after a complete infection cycle: filter onto 5 µm filters with autoclaved devices and wait maturation time according to previous results (18h for A25(ST) and 6h for A120(ST)). Step 3: inoculation of dinospores on a new fresh host culture and collect samples each 15 minutes for 1h and each hour for 3h. We repeated the test with the A25(ST)*ST147 to increase this maturation period (24 hours). Experiment 2: Fate and infectivity of dinospores Next part consisted on collected samples to follow dinospores fate: maturation and infectivity. This protocol was also designed regarding previous FISH results (blogpost from 11/11) and results from a). Step 1 consisted on dinospore production similar to a) but ratio 1:3 to boost infection. Step 2 is proceeded 24h incubation later for reduction of the bacterial contamination. Infected host cells are collected using 10 µm nylon filters, then resuspended into 30 mL of fresh medium (recently filtrated again onto 0.2 µm) twice. Second time, we filter 200mL media to rinse cells. Cells are resuspended into 560 mL of fresh medium. This culture was then split into 6 different flasks (90 mL each). In three of them, antibiotics (PNS: Penicillin-Streptomycin-Neomycin), were added (3% final concentration). Step 3 consists of dinospore production and monitoring. After 24 hours, the culture was checked every 6h for dinospore production. Freshly produced dinospores were gathered by 5 µm PC filtration. Samples were collected for flow cytometry (signatures and ploidy levels) analyses and DAPI microscopy each 2 hours during 22h for A25(ST) and each hour for 8h for A120(ST). At same times, a sample was used to reinfect fresh host cultures at a final ratio of 1:10 (parasite: host) for A25(ST) and A120(ST). Incubation times differ over strains and were determined based on Experiment 1. Cultures were then fixed with PFA (1% final concentration) for FISH analyses.
0 Comments
Leave a Reply. |