|
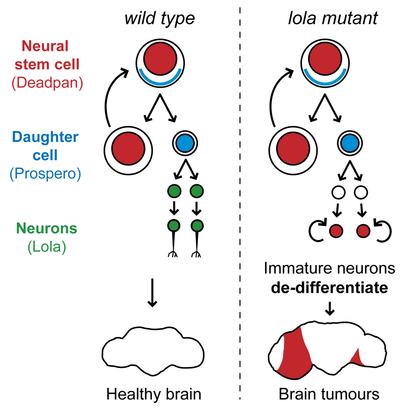
Complex organisms as we are rely on the differentiation of stem cell to form muscle, blood, immune cells and... neurons. During nervous system development, neural stem cells asymmetrically divide and give rise to two daughter cells. A new neural stem cells ensures self-renewal. Ganglion Mother cells (GCM) then further differentiate into neurons and glial cells to fulfill specialized functions. Which factor control the choice between neural stem cell renewal and differentiation? Stem cells are carrying the same genetic information but the regulation of gene expression determines the outcome of a division. This highly ordered activation of repression of genes results in modifications of cell’s shape, size and metabolic activity. The Southall lab , where I am currently doing my internship, has investigated the factors determining the fate of neural stem cells, using Drosophila melanogaster as a model organism (reference for the original research paper below). The transcription factor Deadpan is expressed in the nucleus of initial neural stem cell to drive cell division. At this stage, another TF, Prospero, is expressed in the cytosol of one side of the cell. If it enters the nucleus, it represses genes required for self-renewal and activates genes for neuronal and glial cell differentiation. So, Prospero determines the choice between self-renewal and differentiation. In prospero mutant, GMC effectively failed to differentiate and they reverted to stem cells characteristics. Deadpan is not repressed anymore, cells start dividing, and eventually tumors can be initiated. Because Prospero appeared to both activate and repress gene expression, it was likely that some co-factor was collaborating to modulate its activity. The sequence which is bound by Prospero binding has been extracted and many other proteins which recognized this motif were identified. However, a yeast one hybrid assay, by which DNA-protein interaction can be assessed, has revealed Lola-N, a specific isoform from the 25 splice variants of the BTB-zinc finger transcription factor Lola. So, Lola, a transcription factor, seems to bind the same molecular targets as Prospero. Are they acting together? When and where is Lola expressed? A widespread method used to answer this question is the staining with antibodies, which label proteins with fluorescent tags. Lola appeared to be expressed in post-mitotic neuron, after GMC division and Prospero expression. So, the brief expression overlap between Lola and Prospero does not ague for their collaboration. How does Lola act in postmitotic neuron? Lola's binding site and interactions with its targets were identified. Lola represses several neural stem cells genes such as deadpan and other genes initiating cell cycle. That is why it is called a tumor suppressor. However, Prospero can repress neural stem cells genes as well! Because Lola expression level only increases at the final division of GMC and then in post mitotic neurons, it is likely that Prospero initiates cell differentiation while Lola is required to maintain newly formed neuron in a differentiated state. As expected, in lola mutant, removing Lola resulted in the dedifferentiation of immature neurons, which began to divide and form tumors. Therefore, although it was well-known that cells could be reprogrammed and re-differentiated into the desired lineage, these results provided new information on factors required to prevent dedifferentiation. On the other hand, Lola removal does not cause dedifferentiation of mature neurons… More permanent mechanisms might be recruited later in the development. So, something might bind Lola to regulate its function… My aim for this project is to investigate one of the factor likely regulating Lola’s function and I’ll explain how in my next blog post... A perspective for regenerative medicine? Lola prevents immature neurons from dedifferentiating … imagine if we could induce neurons to dedifferentiate, and then direct their differentiation to neurons of our choice, it would be an amazing method of repair of diseased tissues in vivo! Even more since the same factor can maintain repression of neural stem cells genes, avoiding cell division and gliomas… Useful links: Southall, T., Davidson, C., Miller, C. & Carr, A. et al. (2014) Dedifferentiation of Neurons Precedes Tumor Formation in lola Mutants. Developmental Cell. [Online] 28 (6), 685-696. Available from: doi:10.1016/j.devcel.2014.01.030. Only 1 minute to understand neural stem cells? Watch this video!
6 Comments
Noémie Paillon
15/3/2017 03:14:42 am
Hello Cécile !!
Reply
Cécile Crapart
15/3/2017 10:21:56 am
Thanks for your questions because I learned more about Prospero asymetrical disctibution during mitosis. Another protein, Miranda, is required for the repartition of Prospero between the new neuroblast and the GMC:
Reply
Noémie Paillon
16/3/2017 02:11:15 am
Ok thank you ! If I understood well, miranda anchors Prospero to the membrane, so I don't think it's required for Prospero's translocation in the GMC's nucleus because “in embryos homozygous for the deficiency of miranda, […] Prospero stays in the cytoplasm during mitosis and is translocated into the nuclei of both daughter cells”. Miranda seems to be required only for its anchoring action, but the translocation into the nucleus might be initiated by an NLS.
Cécile
16/3/2017 08:04:37 am
Yes I think you are right for Miranda's function, in the paper form Cell tey say : "Miranda and Prospero are colocalized during mitosis. They form crescents that are associated with the basal cell membrane (fig 3 A and B). At the end of telophase, they are segregated into the GMC, the basal daughter (C and D). Shortly after cell division in the GMC, Prospero is released from the membrane and translocated into the nucleus (F), whereas Miranda becomes undetectable (E)."
Reply
margaux
21/3/2017 03:56:45 am
I read the word "gliomas" and here I come
Reply
Cécile
22/3/2017 08:45:03 am
Well, that's a very good question. But I think this will not be used as a preventative method but more in the context of experiments with stem cells. I think that, such as factors necessary to reprogram differentiated cells into induced pluripotent stem cells and then direct their differentiation have been identified, informations about Lola could help any studies aiming to differentiate stem cells into neurons and promote maintenance/avoid dedifferentiation.
Reply
Leave a Reply. |