|
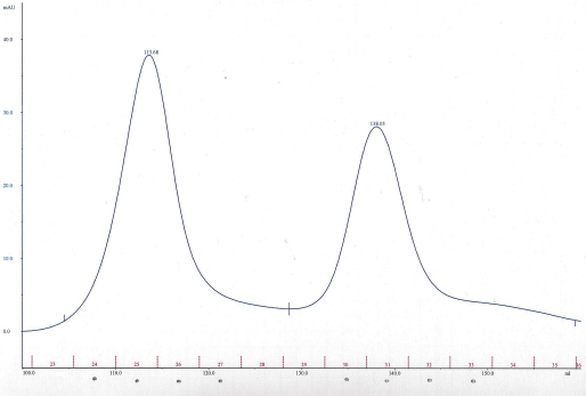
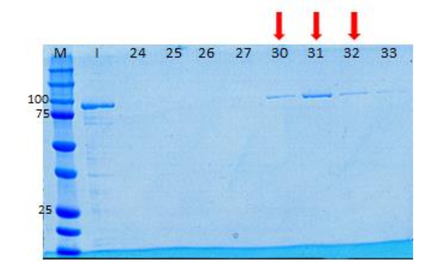
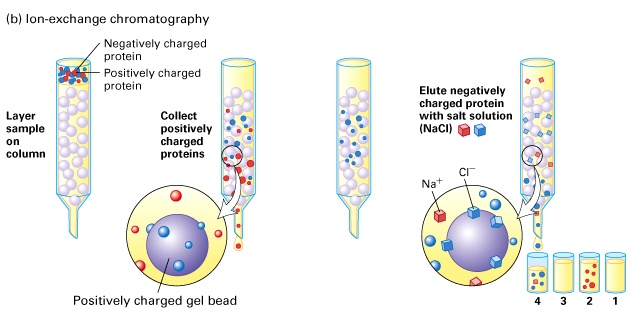
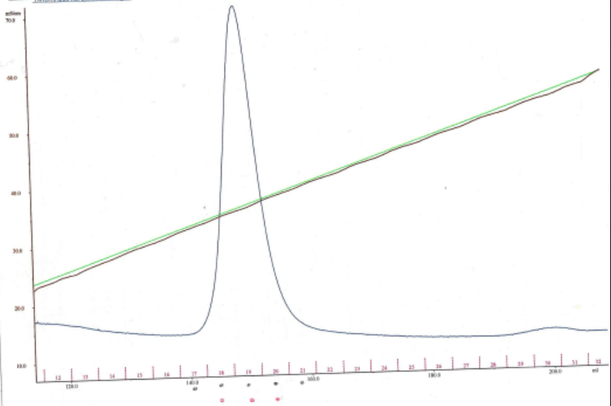
My aim then was to purify Hsp90 at a larger scale. To do so, I followed a six-step protocol. Here are the six steps : - Cell lysis - HisTrap affinity chromatography - Gel filtration chromatography - Anion exchange chromatography - ATP-agarose affinity chromatography - Storage of the purified protein It will be very long to discuss about all of these steps so I will simply talk about cell lysis, gel filtration chromatography, anion exchange chromatography and storage of the purified protein. Cell lysis This time, to lyse the cells I used sonication.  I pulled out my bacterial pellet from the freezer (-20°C) and I suspended it in a buffer solution. I placed my sample in an ice bath and I sonicated my bacterial suspension for 1 min then I paused for 1 min. I repeated this 4 times. Then, I centrifuged the obtained total lysate at 30000 rcf (relative centrifugal force) during 30 min. After that, I sampled the supernatant (soluble lysate) that I kept for the next steps. A sonicator. source : https://commons.wikimedia.org/wiki/File:Sonicator_togopic.png Gel filtration chromatography After HisTrap affinity chromatography, we mixed all the fractions that contained the protein and we got a final volume of 5mL. It's that sample of 5mL that I used for gel filtration chromatography. 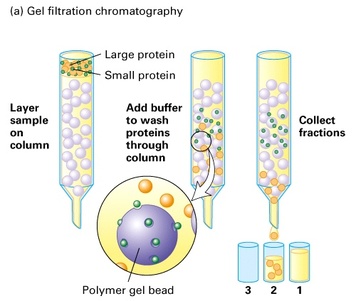 Gel filtration chromatography which is also called size exclusion chromatography separates molecules according to the size. Gel filtration resin can be thought as beads having little pores. Large proteins which cannot enter these pores will pass around the beads whereas small proteins will flow through the beads and thus take a longer path. The larger proteins will therefore elute first and the smallest ones will elute last. source : http://www.pha.jhu.edu/~ghzheng/old/webct/note1_1.htm Here is the graph that I obtained after gel filtration chromatography : The pics indicate the presence of proteins. To know if my protein of interest was collected in the fractions 24, 25, 26 and 27 (see black dots under the first pic) or in the fractions 30, 31, 32 and 33 (see black dots under the second pic), I did an SDS-PAGE gel. The presence of distinct bands around 90kDa (see red arrows) proved me that Hsp90 was in the fractions 30, 31 and 32. Anion exchange chromatography We mixed these three fractions and we got a final volume of 15mL. It's that sample of 15mL that I used for anion exchange chromatography. source : http://www.pha.jhu.edu/~ghzheng/old/webct/note1_1.htm In the anion exchange chromatography, negatively charged proteins will bound to the positively charged beads whereas positively charged proteins will pass through the column. Therefore, positively charged proteins will elute first. Negatively charged proteins are eluted with a salt solution. Here is the graph that I obtained after anion exchange chromatography : I collected the fractions 18, 19 and 20 (see red dots). After mixing these fractions, I got a final volume of 15mL. It's that sample of 15mL that I used for the next step. Storage of the purified protein
After ATP-agarose affinity chromatography, I concentrated the eluate and I obtained a volume around 180µL. After measuring the concentration of this aliquot of 180µL, we found that our protein was at a very low concentration. Indeed, we get six aliquots of 30µL concentrated at 1.23mg/mL. We kept these six aliquots at -80°C.
2 Comments
Manon
15/11/2016 04:28:44 am
Hi Maryam!
Reply
Cécile
16/11/2016 01:46:51 pm
Hey!
Reply
Leave a Reply. |